Non‐Chelate‐Assisted Palladium‐Catalyzed Aerobic Oxidative Heck Reaction of Fluorobenzenes and Other Arenes: When Does the C−H Activation Need Help? - ScienceDirect

Single palladium atoms stabilized by β-FeOOH nanorod with superior performance for selective hydrogenation of cinnamaldehyde | SpringerLink

Catalytic Activity of Bimetallic (Ruthenium/Palladium) Nano‐alloy Decorated Porous Carbons Toward Reduction of Toxic Compounds - Veerakumar - 2019 - Chemistry – An Asian Journal - Wiley Online Library
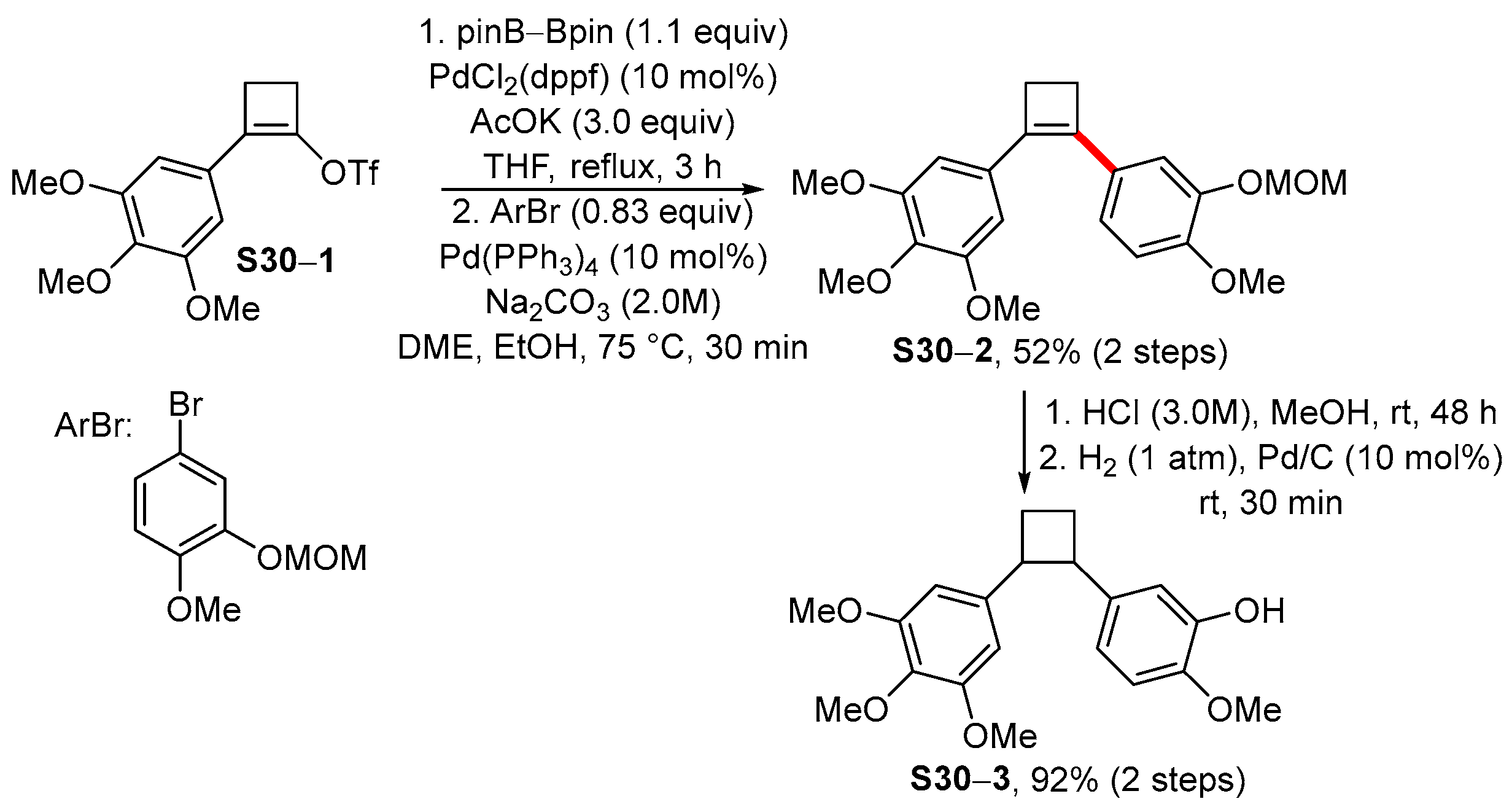
Organics | Free Full-Text | Palladium-Catalyzed Cross-Coupling Reactions of Borylated Alkenes for the Stereoselective Synthesis of Tetrasubstituted Double Bond | HTML

C–H Bond Activation Mechanism by a Pd(II)–(μ-O)–Au(0) Structure Unique to Heterogeneous Catalysts | JACS Au

Palladium in biological media: Can the synthetic chemist's most versatile transition metal become a powerful biological tool? - ScienceDirect
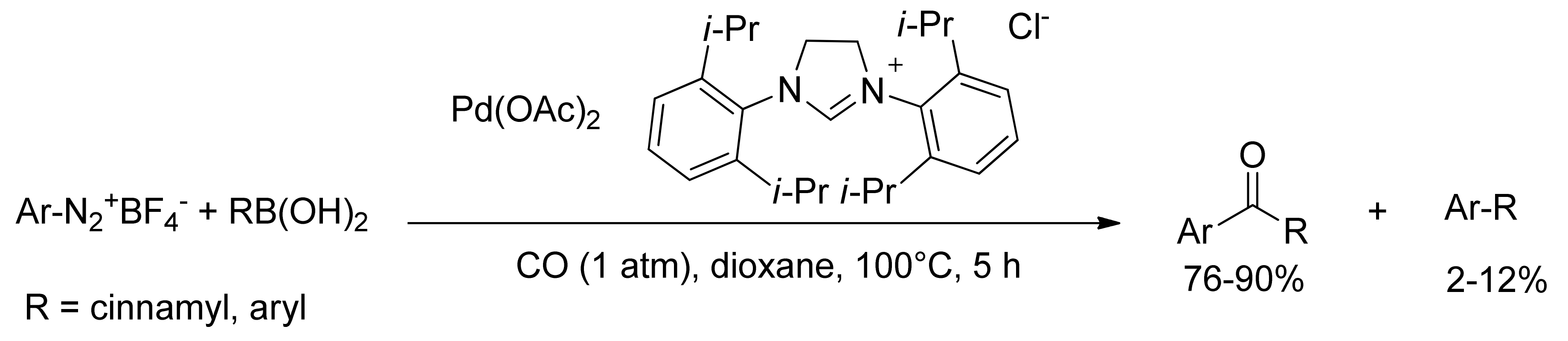
Molecules | Free Full-Text | Suzuki-Miyaura Cross-Coupling in Acylation Reactions, Scope and Recent Developments | HTML

Palladium-Catalyzed Synthesis of Functionalized Indoles by Acylation/Allylation of 2-Alkynylanilines with Three-Membered Rings | Organic Letters

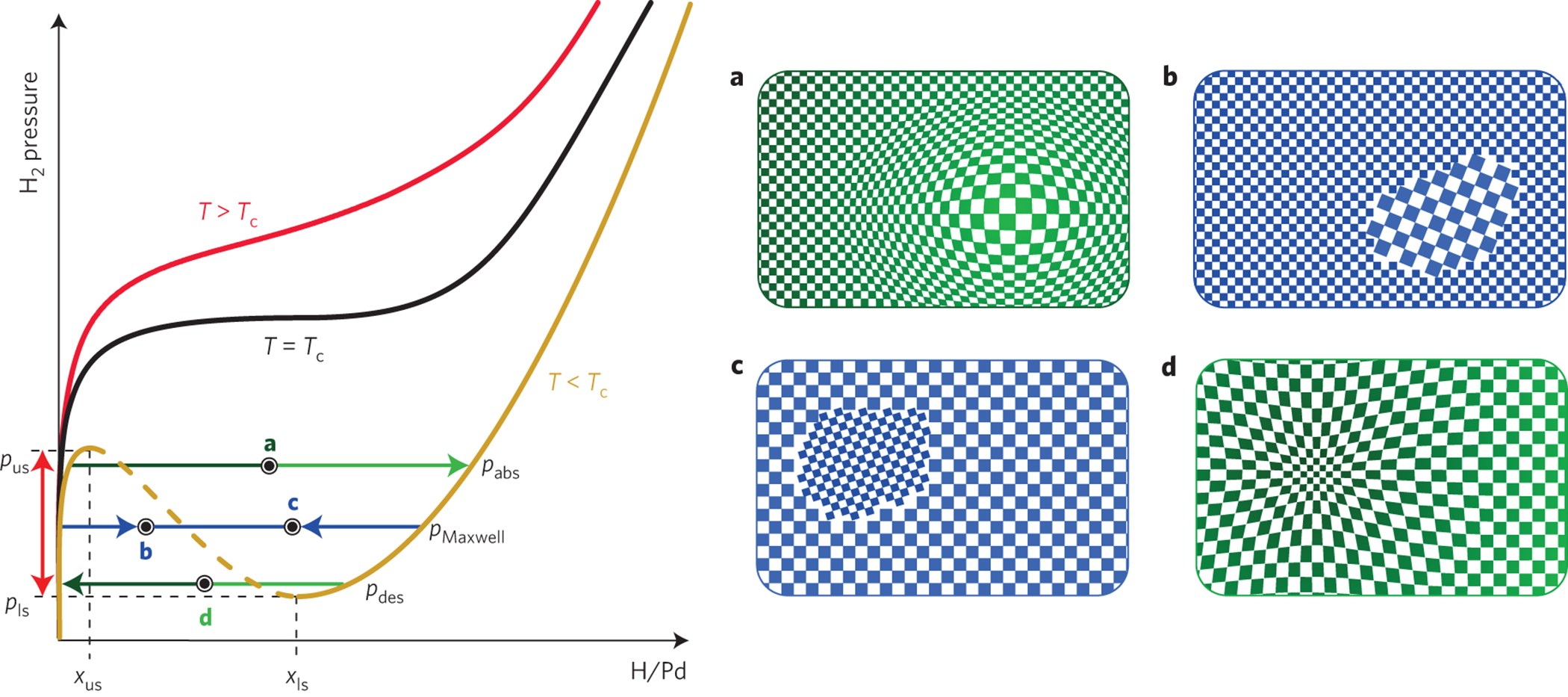
High-Performance Nanostructured Palladium-Based Hydrogen Sensors—Current Limitations and Strategies for Their Mitigation | ACS Sensors

Combined Effect of Palladium Catalyst and the Alcohol to Promote the Uncommon Bis‐Alkoxycarbonylation of Allylic Substrates - Olivieri - 2022 - ChemCatChem - Wiley Online Library

Palladium-Catalyzed Enantioselective β-C(sp3)–H Activation Reactions of Aliphatic Acids: A Retrosynthetic Surrogate for Enolate Alkylation and Conjugate Addition | Accounts of Chemical Research

Recent Development in Palladium-Catalyzed Domino Reactions: Access to Materials and Biologically Important Carbo- and Heterocycles | Organometallics

Understanding the Relative Easiness of Oxidative Addition of Aryl and Alkyl Halides to Palladium(0) | Organometallics