
Scheme 14 | Synthesis of highly functionalized organic compounds through Ugi post-transformations started from propiolic acids | SpringerLink

3,5-Diiodothyronine in vivo maintains euthyroidal expression of type 2 iodothyronine deiodinase, growth hormone, and thyroid hormone receptor β1 in the killifish | American Journal of Physiology-Regulatory, Integrative and Comparative Physiology

PDF) Multifunctional Deuterated and Tritiated 'Click' Molecular Probes via Palladium-Mediated Reductive Deiodination of 5-Iodo-1,2,3-Triazoles | Eva Lenz - Academia.edu

Palladium‐Catalyzed Selective meta‐C−H Deuteration of Arenes: Reaction Design and Applications - Bag - 2019 - Chemistry – A European Journal - Wiley Online Library
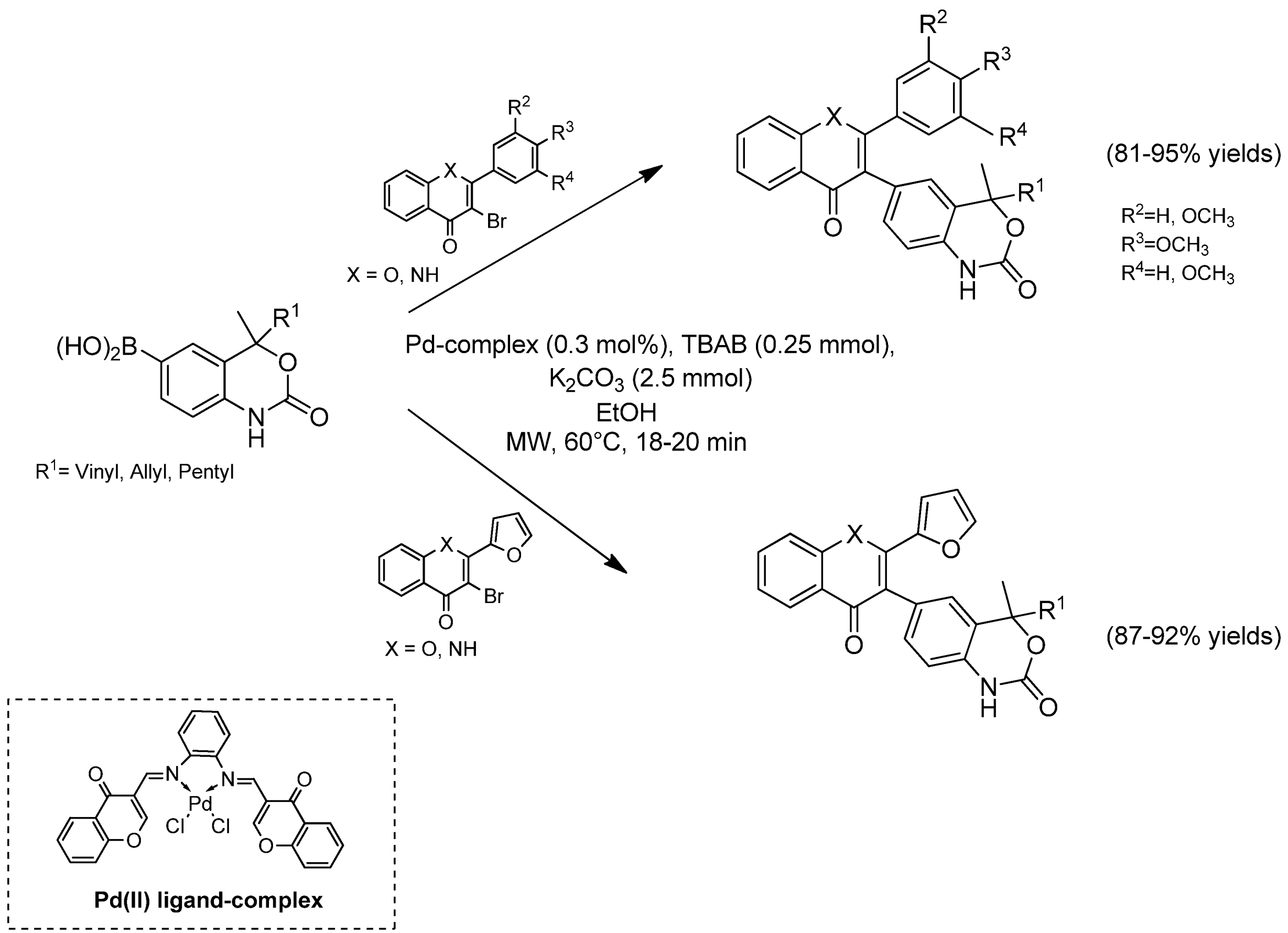
Catalysts | Free Full-Text | Eco-Friendly Physical Activation Methods for Suzuki–Miyaura Reactions | HTML

Electrochemical dehalogenation of disinfection by-products and iodine-containing contrast media: A review - ScienceCentral

Transition-Metal (Pd, Ni, Mn)-Catalyzed C-C Bond Constructions Involving Unactivated Alkyl Halides and Fundamental Synthetic Building Blocks. - Abstract - Europe PMC

Pd concentration in the supernatant after centrifugation (8041 g for 7... | Download Scientific Diagram
Electrochemical dehalogenation of disinfection by-products and iodine-containing contrast media: A review
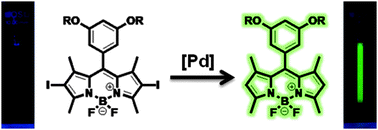
A fluorescence turn-on sensor for the detection of palladium ions that operates through in situ generation of palladium nanoparticles - Chemical Communications (RSC Publishing)
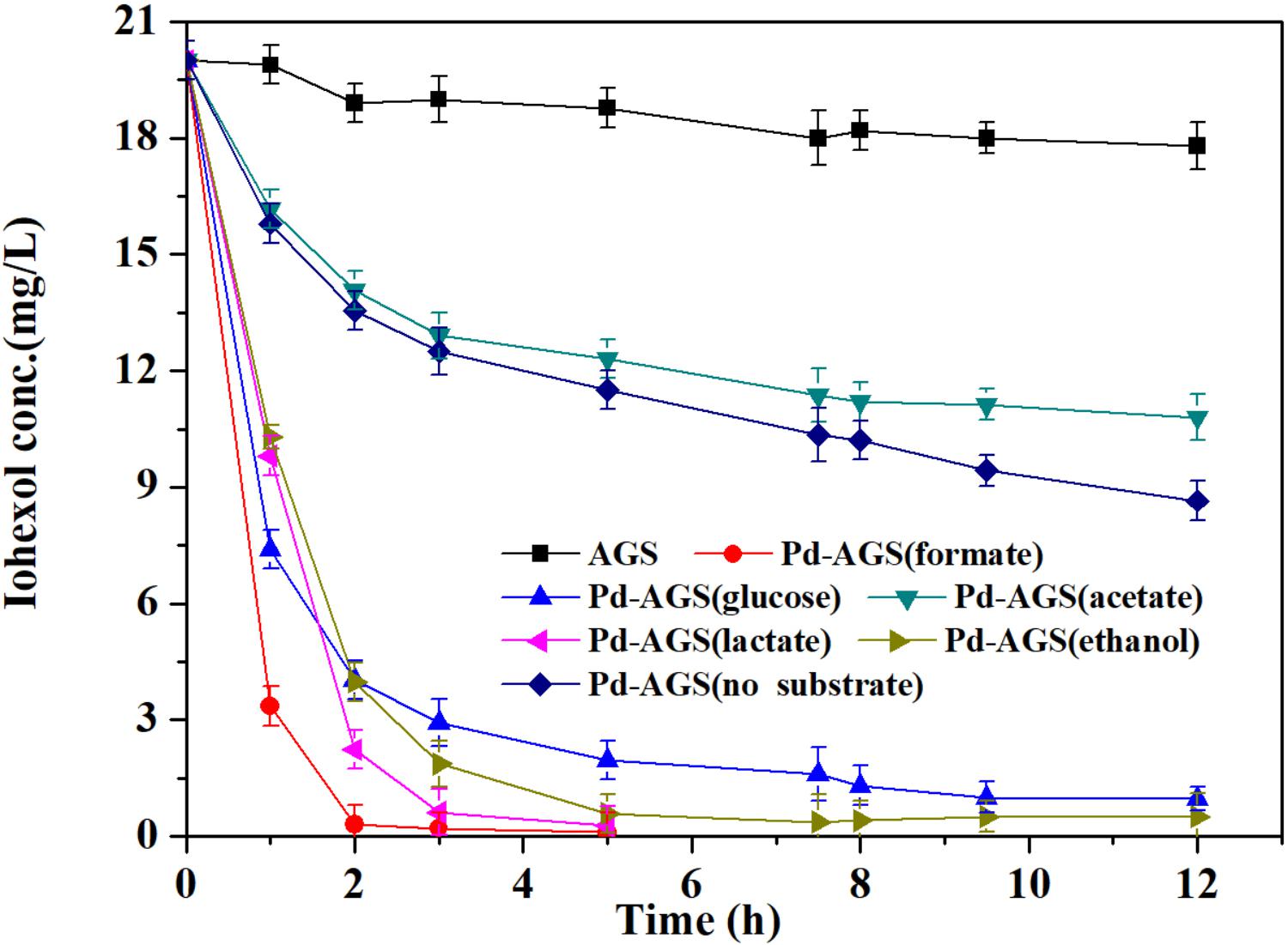
Frontiers | Iohexol Degradation by Biogenic Palladium Nanoparticles Hosted in Anaerobic Granular Sludge | Microbiology

3,5-Diiodothyronine in vivo maintains euthyroidal expression of type 2 iodothyronine deiodinase, growth hormone, and thyroid hormone receptor β1 in the killifish | American Journal of Physiology-Regulatory, Integrative and Comparative Physiology

PDF) A naked-eye and ratiometric near-infrared probe for palladium via modulating π-conjugated system of cyanines

From Benzofurans to Indoles: Palladium‐Catalyzed Reductive Ring‐Opening and Closure via β‐Phenoxide Elimination - Perego - 2019 - Advanced Synthesis & Catalysis - Wiley Online Library
Electrochemical dehalogenation of disinfection by-products and iodine-containing contrast media: A review
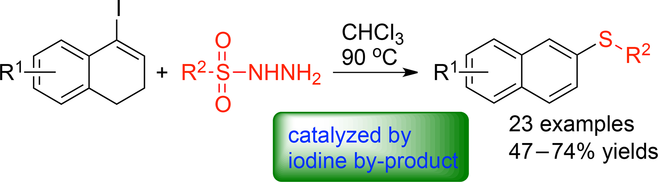
By‐Product‐Catalyzed Redox‐Neutral Sulfenylation/Deiodination/Aromatization of Cyclic Alkenyl Iodides with Sulfonyl Hydrazides,Advanced Synthesis & Catalysis - X-MOL








