Pd 2+ coordination induced fluorescence quenching of selenium-donor... | Download Scientific Diagram
![A selenium-coordinated palladium(ii) trans-dichloride molecular rotor as a catalyst for site-selective annulation of 2-arylimidazo[1,2-a]pyridines - Chemical Communications (RSC Publishing) A selenium-coordinated palladium(ii) trans-dichloride molecular rotor as a catalyst for site-selective annulation of 2-arylimidazo[1,2-a]pyridines - Chemical Communications (RSC Publishing)](https://pubs.rsc.org/image/article/2020/cc/d0cc03599h/d0cc03599h-s2_hi-res.gif)
A selenium-coordinated palladium(ii) trans-dichloride molecular rotor as a catalyst for site-selective annulation of 2-arylimidazo[1,2-a]pyridines - Chemical Communications (RSC Publishing)
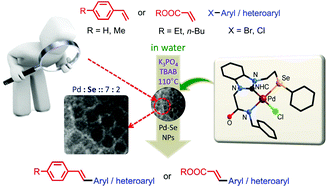
Palladium(ii) ligated with a selenated (Se, CNHC, N−)-type pincer ligand: an efficient catalyst for Mizoroki–Heck and Suzuki–Miyaura coupling in water - Organic & Biomolecular Chemistry (RSC Publishing)

C–Hortho⋯Pd interactions in palladium complexes derived from tetrasulfur-difluorinated ligands. Experimental, computational and catalytic studies - ScienceDirect
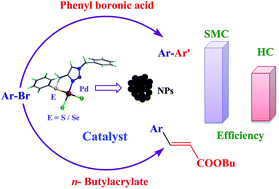
Palladium(ii) complexes bearing the 1,2,3-triazole based organosulfur/ selenium ligand: synthesis, structure and applications in Heck and Suzuki–Miyaura coupling as a catalyst via palladium nanoparticles - RSC Advances (RSC Publishing)

Irradiation/dual ligand enabled palladium-catalysis for decarboxylative desaturation | Nature Research Chemistry Community

Ligand-controlled palladium catalysis enables switch between mono- and di-arylation of primary aromatic amines with 2-halobenzothiazoles - Org. Chem. Front. - X-MOL
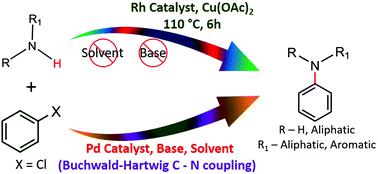
Half-sandwich (η5-Cp*)Rh(iii) complexes of pyrazolated organo-sulfur/ selenium/tellurium ligands: efficient catalysts for base/solvent free C-N coupling of chloroarenes under aerobic conditions. - Org. Biomol. Chem. - X-MOL
![A selenium-coordinated palladium(ii) trans-dichloride molecular rotor as a catalyst for site-selective annulation of 2-arylimidazo[1,2-a]pyridines - Chemical Communications (RSC Publishing) A selenium-coordinated palladium(ii) trans-dichloride molecular rotor as a catalyst for site-selective annulation of 2-arylimidazo[1,2-a]pyridines - Chemical Communications (RSC Publishing)](https://pubs.rsc.org/image/article/2020/cc/d0cc03599h/d0cc03599h-s1_hi-res.gif)
A selenium-coordinated palladium(ii) trans-dichloride molecular rotor as a catalyst for site-selective annulation of 2-arylimidazo[1,2-a]pyridines - Chemical Communications (RSC Publishing)
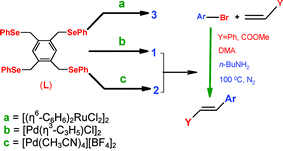
Tetradentate selenium ligand as a building block for homodinuclear complexes of Pd(ii) and Ru(ii) having seven membered rings or bis-pincer coordination mode: high catalytic activity of Pd-complexes for Heck reaction - Dalton
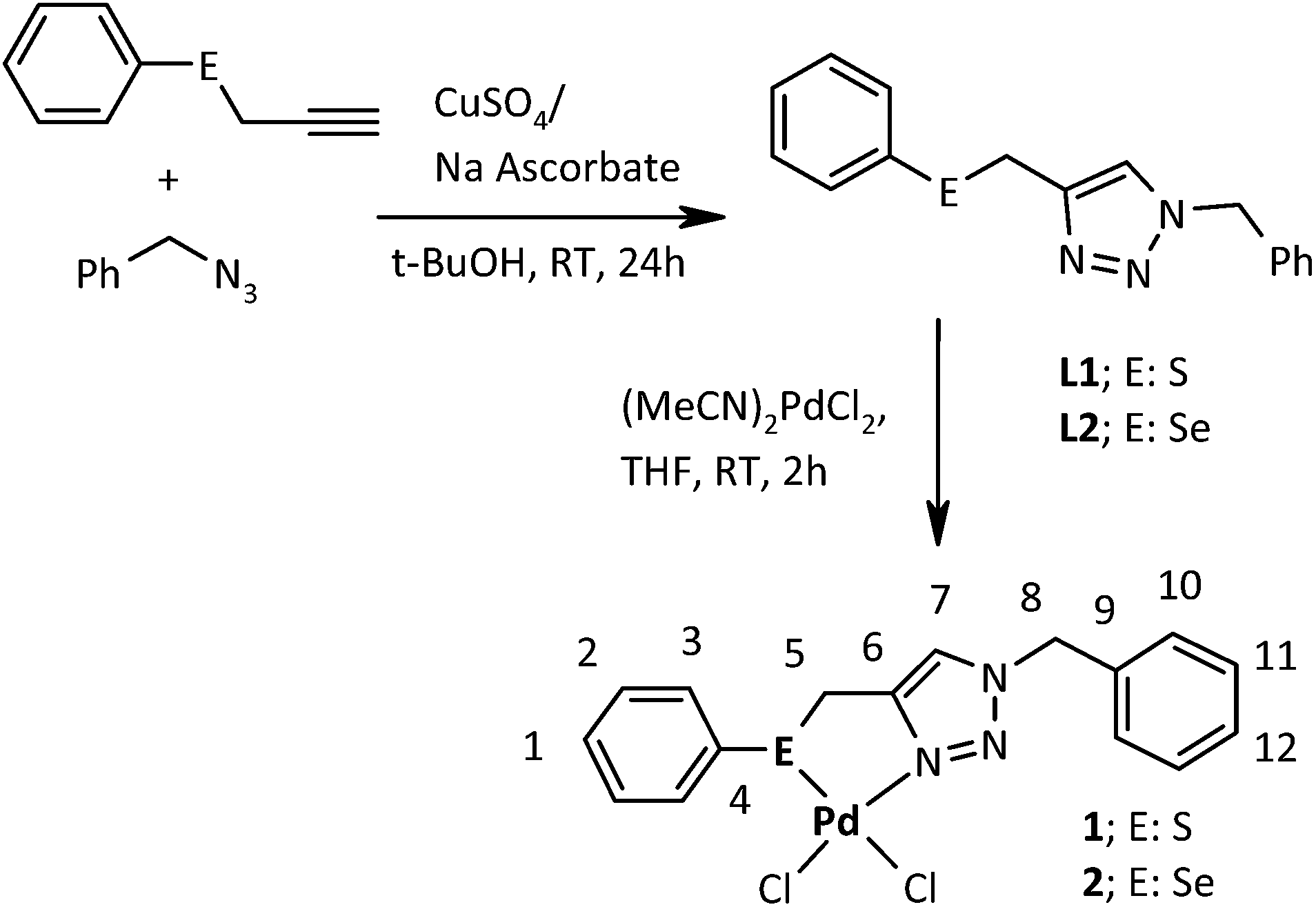
Palladium(ii) complexes bearing the 1,2,3-triazole based organosulfur/ selenium ligand: synthesis, structure and applications in Heck and Suzuki–Miyaura coupling as a catalyst via palladium nanoparticles - RSC Advances (RSC Publishing)

Irradiation/dual ligand enabled palladium-catalysis for decarboxylative desaturation | Nature Research Chemistry Community

ω-Transaminases as efficient biocatalysts to obtain novel chiral selenium-amine ligands for Pd-catalysis - Organic & Biomolecular Chemistry (RSC Publishing)

PDF) Palladacycle containing nitrogen and selenium: highly active pre- catalyst for the Suzuki–Miyaura coupling reaction and unprecedented conversion into nano-sized Pd 17 Se 15 w | Gyaneshwar Rao - Academia.edu
![IUCr) Crystal structure of a palladium(II) complex containing the wide bite-angle bis(selenium) ligand, cis-[(tBuNH)(Se)P(μ-NtBu)2P(Se)(NHtBu)] IUCr) Crystal structure of a palladium(II) complex containing the wide bite-angle bis(selenium) ligand, cis-[(tBuNH)(Se)P(μ-NtBu)2P(Se)(NHtBu)]](https://journals.iucr.org/e/issues/2018/02/00/nk2239/nk2239scheme1.gif)
IUCr) Crystal structure of a palladium(II) complex containing the wide bite-angle bis(selenium) ligand, cis-[(tBuNH)(Se)P(μ-NtBu)2P(Se)(NHtBu)]
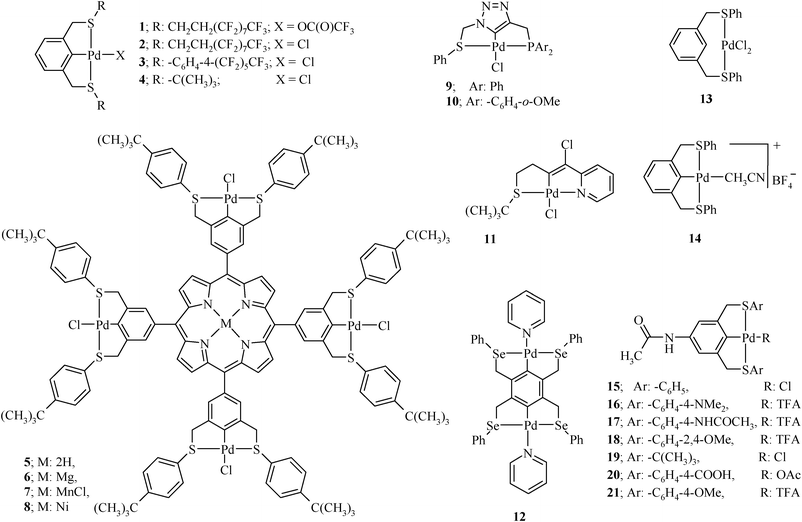
Organochalcogen ligands and their palladium( ii ) complexes: Synthesis to catalytic activity for Heck coupling - RSC Advances (RSC Publishing) DOI:10.1039/C2RA20508D

Full reaction cycle of Pd catalyzed Suzuki reaction using Pd4. Charge... | Download Scientific Diagram

Palladium complexes of chalcogenoethanamine (S/Se) bidentate ligands: Applications in catalytic arylation of CH and OH bonds - ScienceDirect