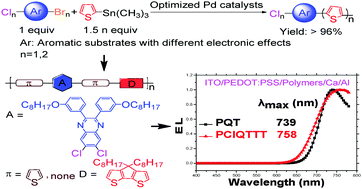
Highly selective palladium-catalyzed Stille coupling reaction toward chlorine-containing NIR electroluminescent polymers - Journal of Materials Chemistry C (RSC Publishing)
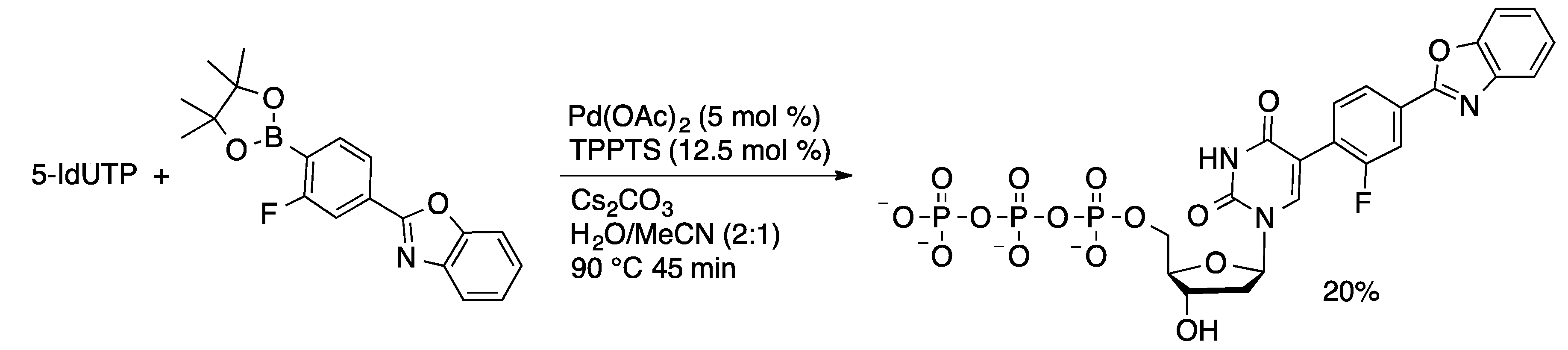
Molecules | Free Full-Text | Palladium-Catalyzed Modification of Unprotected Nucleosides, Nucleotides, and Oligonucleotides | HTML
Molecules | Free Full-Text | Palladium-Catalyzed Modification of Unprotected Nucleosides, Nucleotides, and Oligonucleotides | HTML

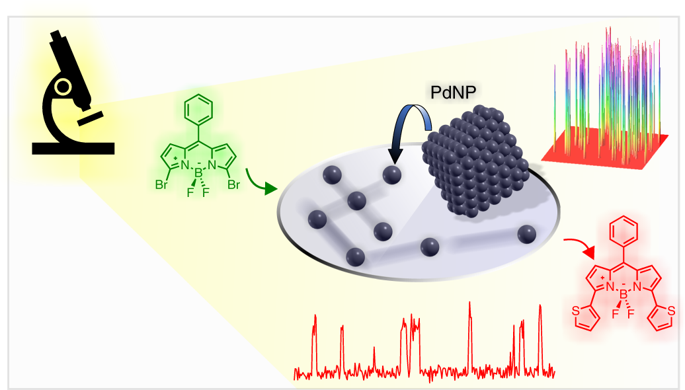
PDF) Palladium-Catalyzed Coupling Reactions for the Functionalization of BODIPY Dyes with Fluorescence Spanning the Visible Spectrum | Taoufik Rohand - Academia.edu
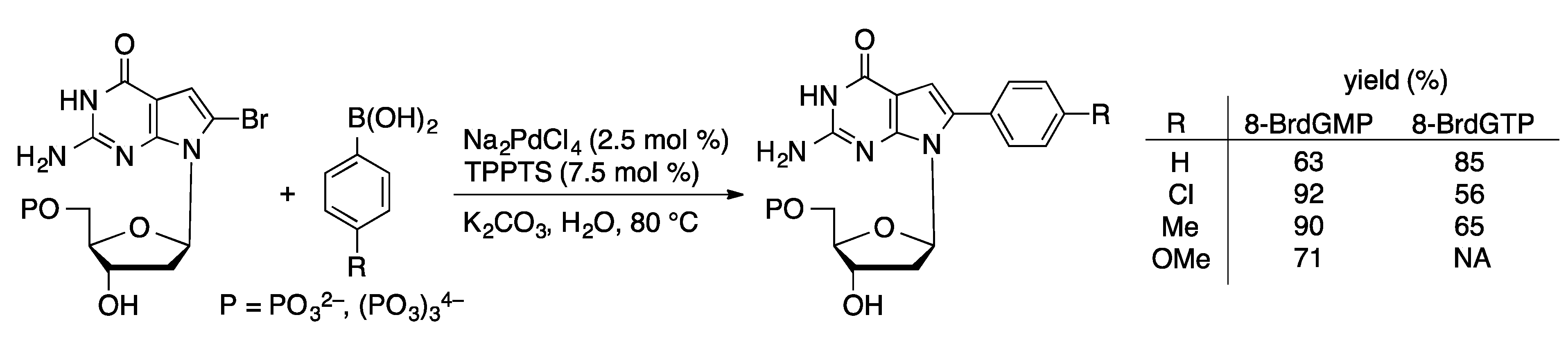
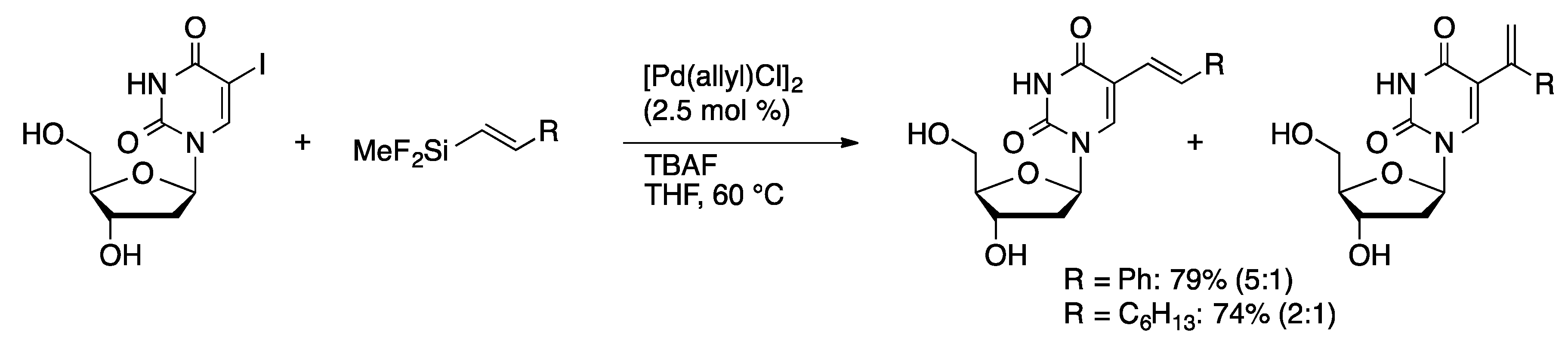
Molecules | Free Full-Text | Palladium-Catalyzed Modification of Unprotected Nucleosides, Nucleotides, and Oligonucleotides | HTML
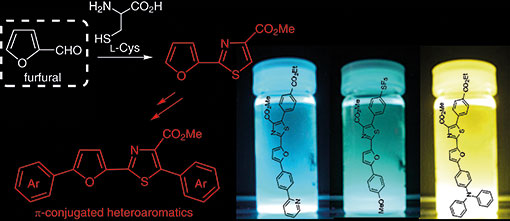
Preparation of Fluorescent Materials from Biomass-Derived Furfural and Natural Amino Acid Cysteine through Cross-Coupling Reactions for Extended π-Conjugation - Synlett - X-MOL

Supramolecular photocatalyst of Palladium (II) Encapsulated within Dendrimer on TiO2 nanoparticles for Photo‐induced Suzuki‐Miyaura and Sonogashira Cross‐Coupling reactions - Eskandari - 2019 - Applied Organometallic Chemistry - Wiley Online Library
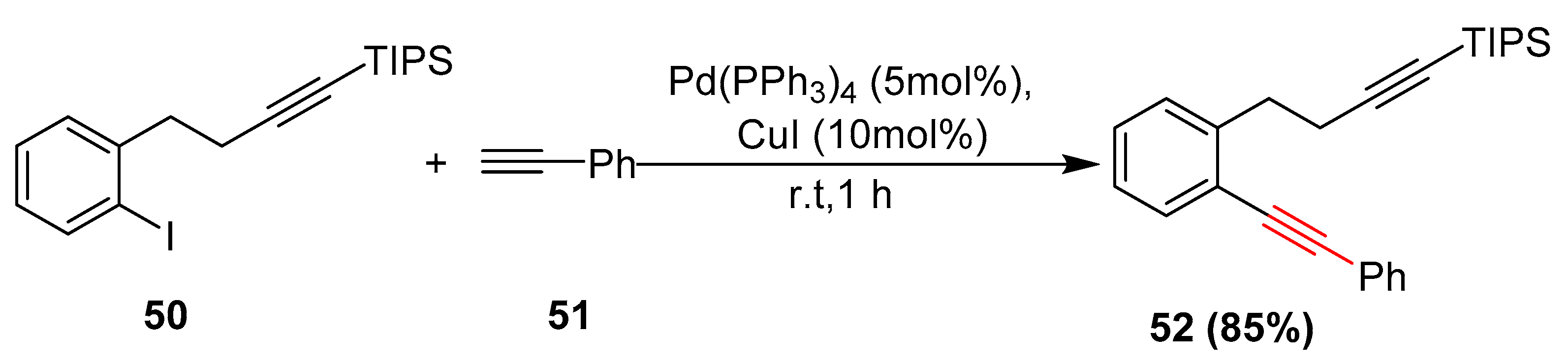
Catalysts | Free Full-Text | Palladium and Copper Catalyzed Sonogashira cross Coupling an Excellent Methodology for C-C Bond Formation over 17 Years: A Review | HTML

3-Phenothiazinyl propiolates – Fluorescent electrophores by Sonogashira coupling of ethyl propiolate - ScienceDirect
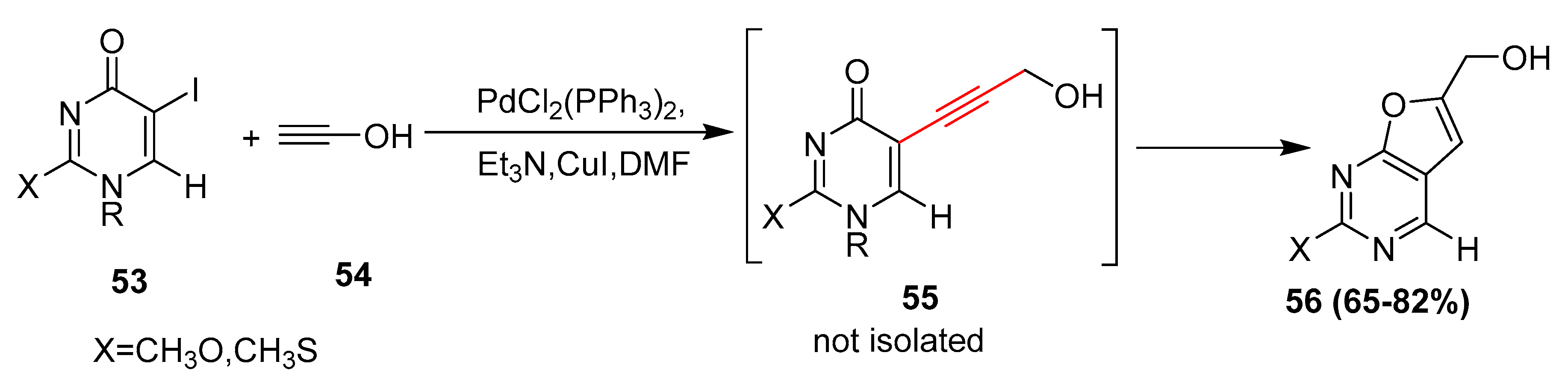
Catalysts | Free Full-Text | Palladium and Copper Catalyzed Sonogashira cross Coupling an Excellent Methodology for C-C Bond Formation over 17 Years: A Review | HTML
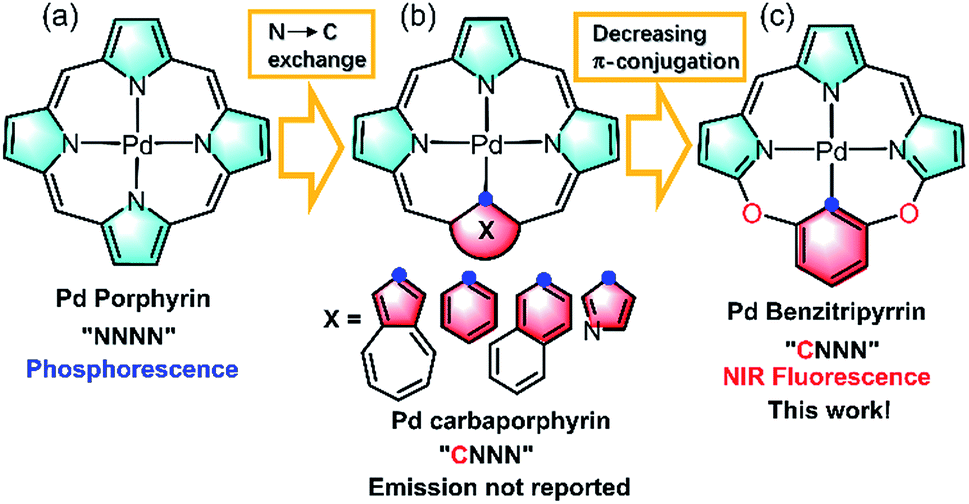
Unusual near infrared (NIR) fluorescent palladium( ii ) macrocyclic complexes containing M–C bonds with bioimaging capability - Chemical Science (RSC Publishing) DOI:10.1039/C9SC04044G

Real-time fluorescence imaging of a heterogeneously catalysed Suzuki–Miyaura reaction | Nature Catalysis

A highly sensitive fluorescence method reveals the presence of palladium in a cross-coupling reaction mixture not treated with transition metals - ScienceDirect

New trends in the cross-coupling and other catalytic reactions in: Pure and Applied Chemistry Volume 89 Issue 10 (2017)
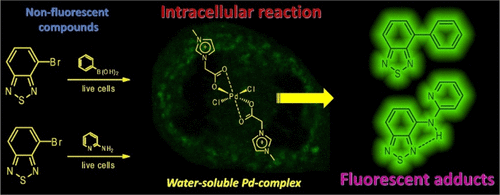
Palladium Catalyst with Task-Specific Ionic Liquid Ligands: Intracellular Reactions and Mitochondrial Imaging with Benzothiadiazole Derivatives. - J. Org. Chem. - X-MOL

Recent Advances in Pd‐Catalyzed Cross‐Coupling Reaction in Ionic Liquids - Li - 2018 - European Journal of Organic Chemistry - Wiley Online Library